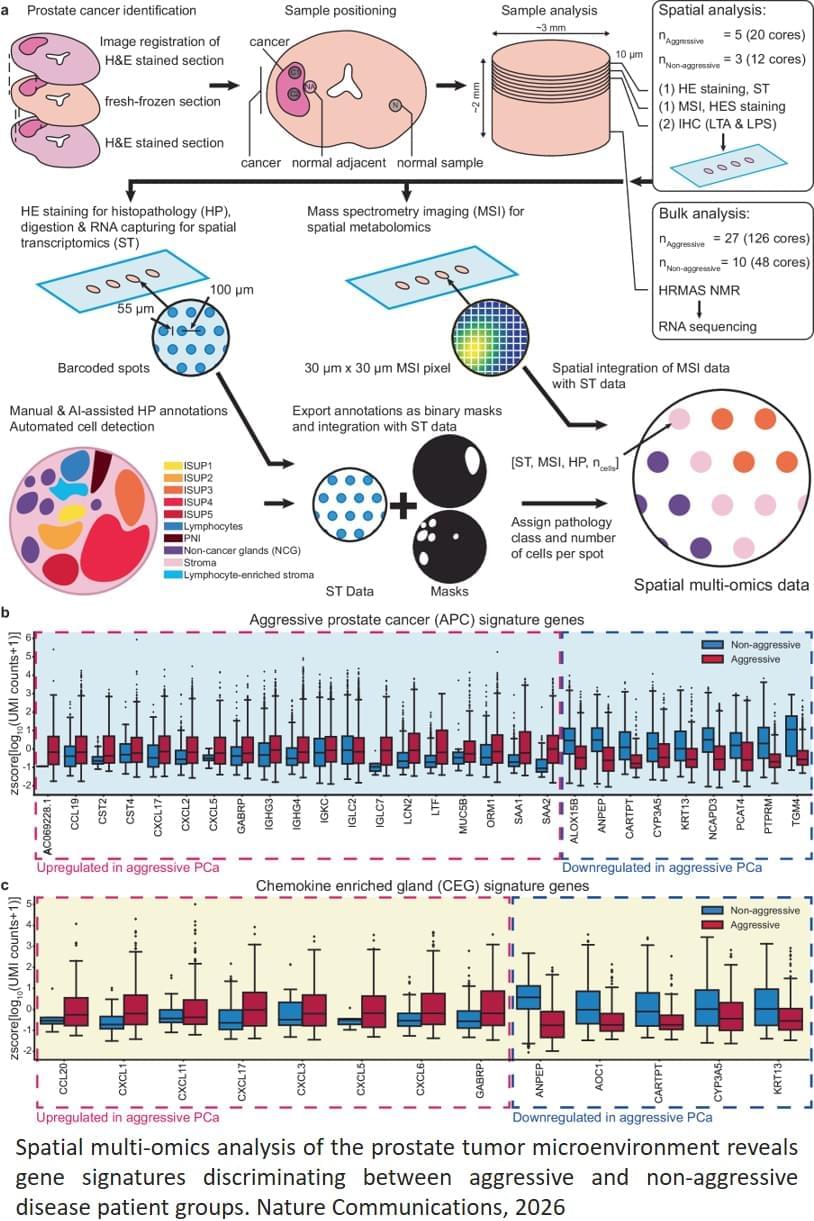
Prostate cancer often develops very slowly. For the vast majority, this is a disease that you live well with, without the need for treatment, but some get an aggressive variant with recurrence of cancer even after surgery. The disease behaves very differently from patient to patient. Understanding what makes the cancer aggressive is crucial for better diagnostics and treatment, says the author.
Aggressive cancer has its own gene expression: The researchers identified a pattern in the gene expression of the tumor itself in prostate tissue in patients with a high risk of recurrence and spread. This signature can become a new tool for distinguishing between patients who need intensive care and those who can manage with less intensive follow-up.
Inflammation of apparently healthy tissue: Signs of inflammation and changes in metabolic processes were also found in the normal tissue close to the cancerous tumor. These glands had high activity of neurotransmitters that attract immune cells, and an increased occurrence of a cell type that can trigger inflammatory reactions. At the same time, the levels of important substances had decreased, suggesting that the gland had lost its normal function.
“Aggressive prostate cancer appears to be associated with inflammation in the area around the cancer cells, combined with specific genetic signatures and metabolic changes in the prostate tissue. This knowledge can provide better methods for early identification of patients at high risk,” says the author. ScienceMission sciencenewshighlights.
The research lays a foundation for the possibility that aggressive prostate cancer can probably be detected through a few drops of semen or blood in the long term.
Prostate cancer is the most common form of cancer among men in Western countries.