Mitochondrial calcium regulates neuronal metabolism and memory.
Brain metabolism is important for long-term memories (LTMs) and various brain functions, Although it is well known that impairing neuronal metabolism limits brain performance, it is not clear if expanding the metabolic capacity of neurons boosts brain function.
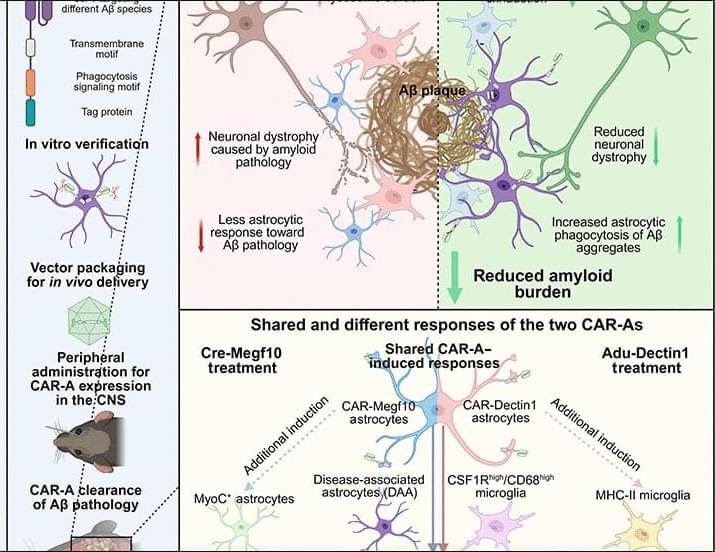
In this study, the authors demonstrate that increasing mitochondrial metabolism can enhance LTM formation in flies and mice.
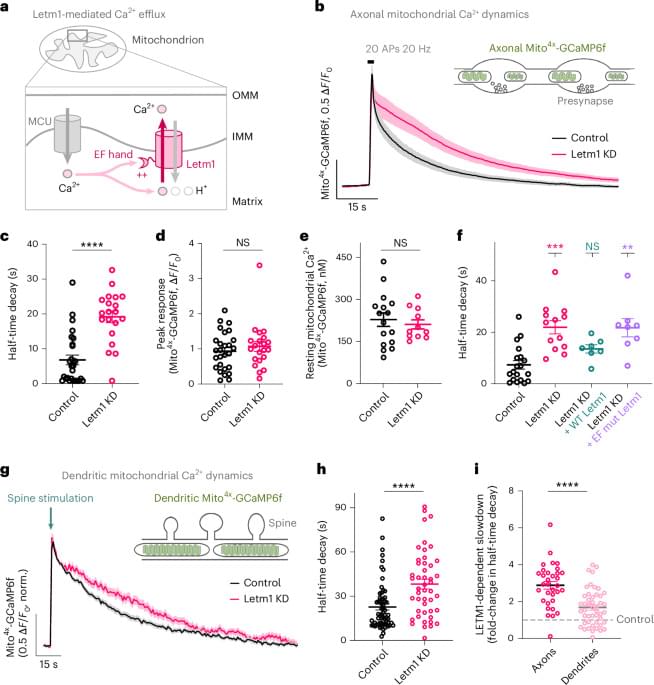
The authors increase mitochondrial Ca2+ by knocking down the mitochondrial Ca2+ exporter Letm1 and demonstrate over-activation of mitochondrial metabolism in neurons of central memory circuits, leading to improved LTM storage. sciencenewshighlights Science Mission https://sciencemission.com/Mitochondrial-Ca2-efflux
Boosting mitochondrial metabolism in neurons in central memory circuits by enhancing Ca2+ retention in the mitochondrial matrix is shown to improve long-term memory formation in flies and mice.